|
Also, of importance, we learned that dapagliflozin's well-established safety profile was consistent in DARE-19."ĪstraZeneca and Saint Luke's Mid America Heart Institute together announced the high-level results of the primary analysis from the trial assessing the potential of Farxiga to treat patients hospitalised with COVID-19 who are at risk of developing serious complications. Mikhail Kosiborod, cardiologist at Saint Luke's Mid America Heart Institute, Vice President of Research at Saint Luke's Health System, and principal investigator of DARE-19, said, "While the trial did not achieve statistical significance, the findings are very interesting and valuable, and will inform future clinical science. The trial also failed to achieve statistical significance for the primary endpoint of recovery measuring a change in clinical status, i.e., from early recovery to death, at 30 days. Compare prices, print coupons and get savings tips for Farxiga (Dapagliflozin) and other Kidney Disease, Heart Failure, and Diabetes Type 2 drugs at CVS, Walgreens, and other pharmacies.  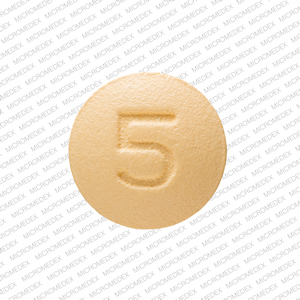
(RTTNews) - British drug major AstraZeneca Plc (AZN, AZN.L) Monday announced that its DARE-19 Phase III trial for Farxiga (dapagliflozin) in COVID-19 did not achieve statistical significance for the primary endpoint of prevention measuring organ dysfunction and all-cause mortality.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
